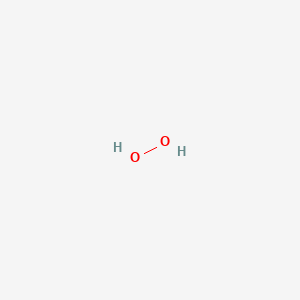
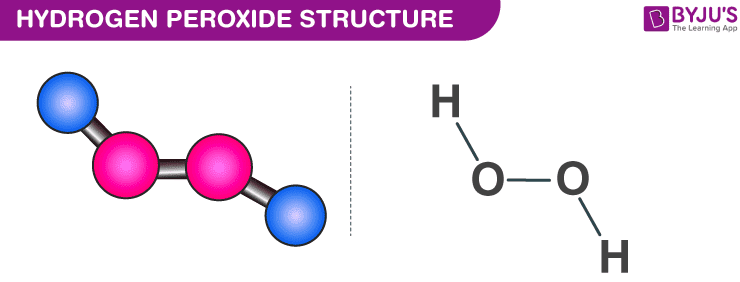
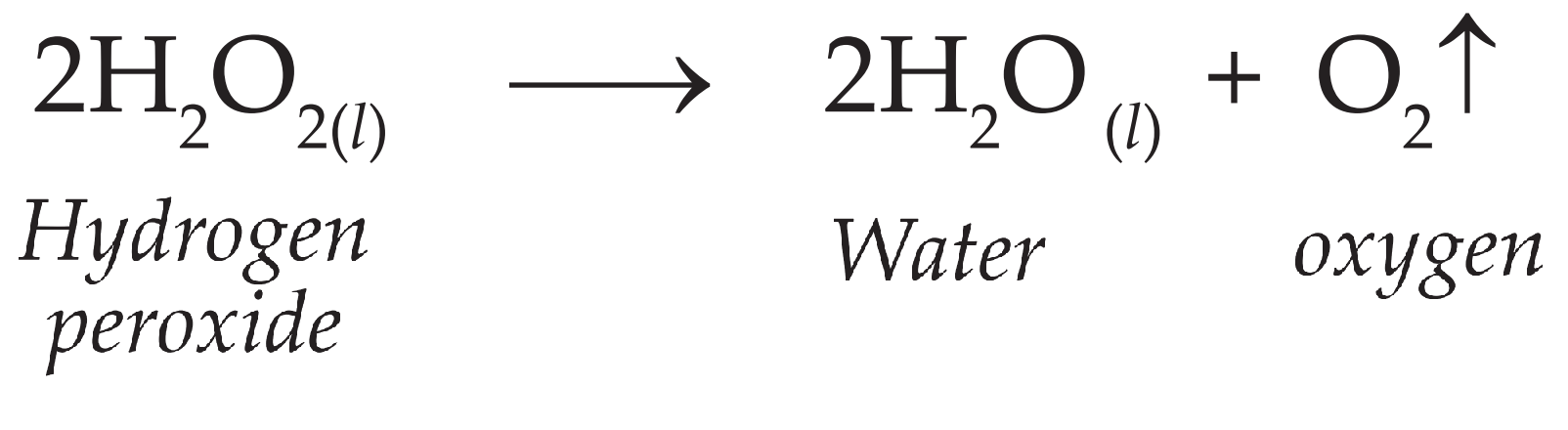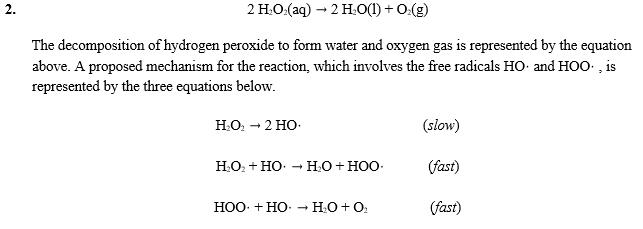Decomposition reaction of H2O2 over Pd/C catalyst in an aqueous medium at high pressure: Detailed kinetic study and modelling - ScienceDirect
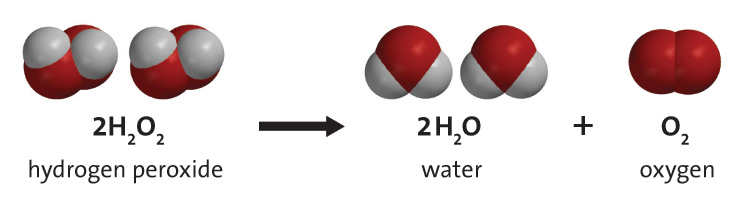
How does a catalyst make Hydrogen Peroxide's decomposition quicker? What is actually happening? | Socratic

SOLVED:The decomposition of hydrogen peroxide is catalyzed by iodide ion. The catalyzed reaction is thought to proceed by a two-step mechanism: H2 O2(a q)+I^-(a q) ⟶H2 O(l)+IO^-(a q) (slow) IO^-(a q)+H2 O2(a q)
![PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f2948b1ccb4f9f878523a741f22c7a299fa747b2/2-Figure1-1.png)
PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar

OneClass: A proposed mechanism for the decomposition of hydrogen peroxide consists of three elementar...
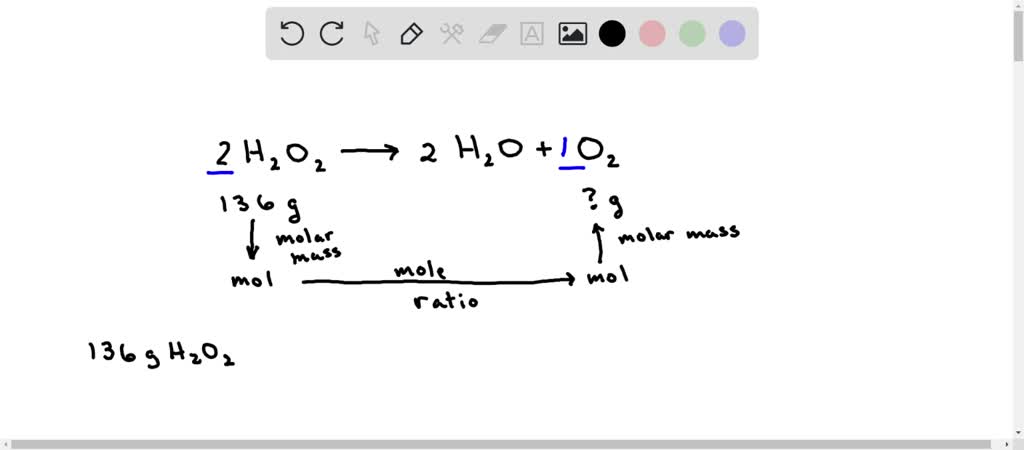
SOLVED: According to the balanced equation below describing the decomposition of hydrogen peroxide to form water and oxygen, if 136 grams of hydrogen peroxide are used, what mass of oxygen gas will


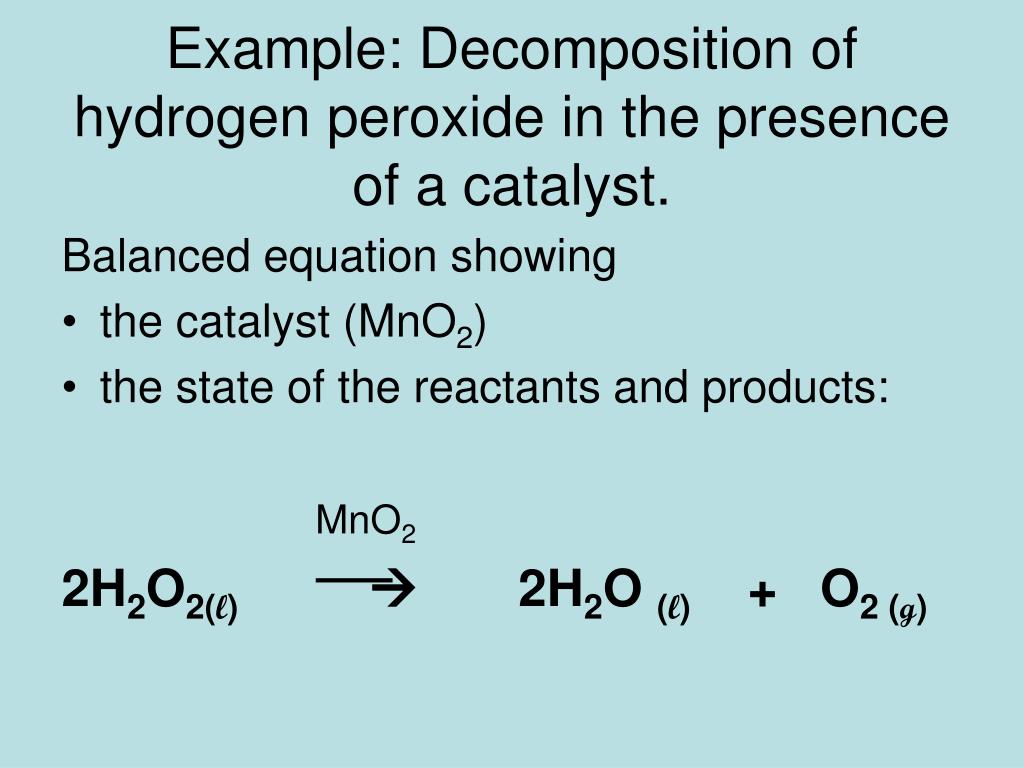
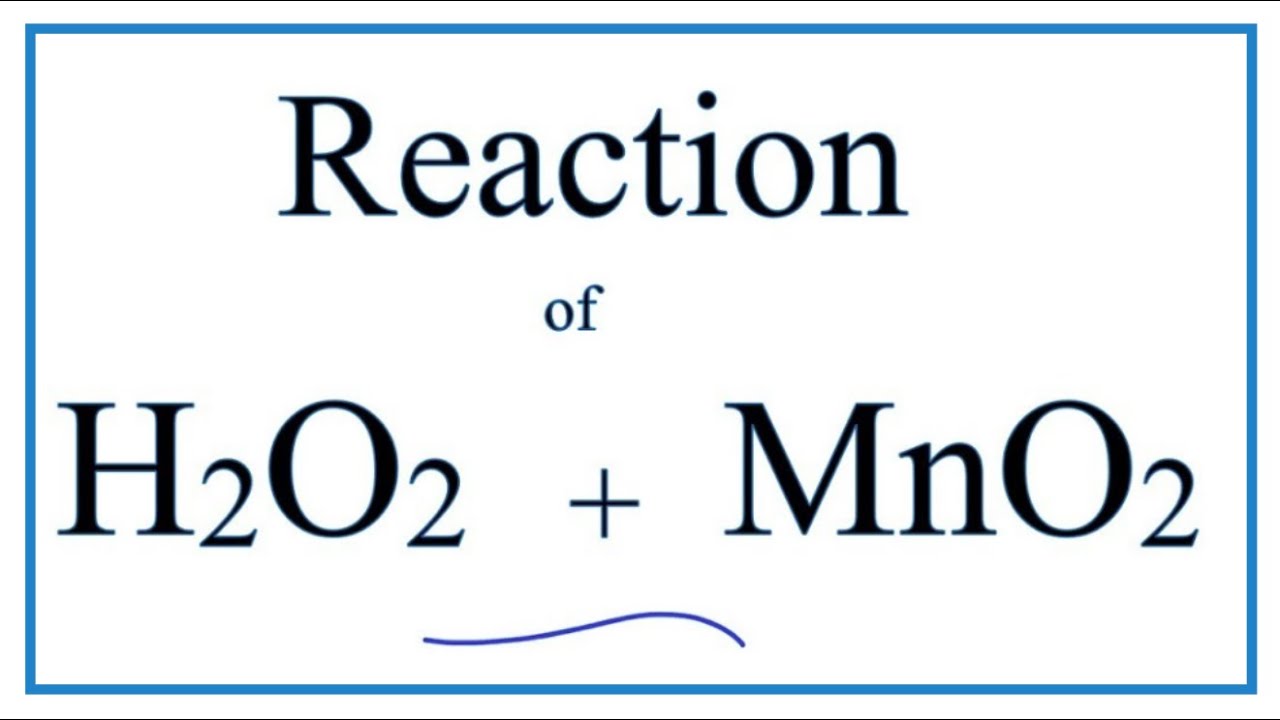



![For the reaction ; 2H2O2(aq)→ 2H2O(l) + O2(g) , rate of decomposition for H2O2 = k[H2O2]^2 For the reaction ; 2H2O2(aq)→ 2H2O(l) + O2(g) , rate of decomposition for H2O2 = k[H2O2]^2](https://dwes9vv9u0550.cloudfront.net/images/2785739/3a8762db-6023-4a5c-9588-862513a5c19d.jpg)